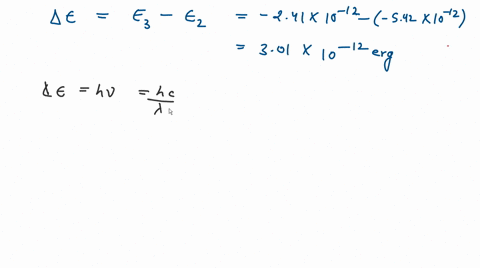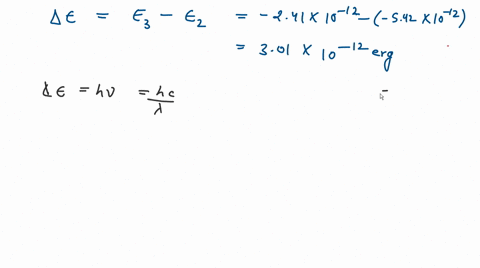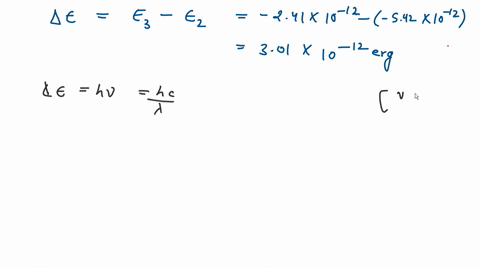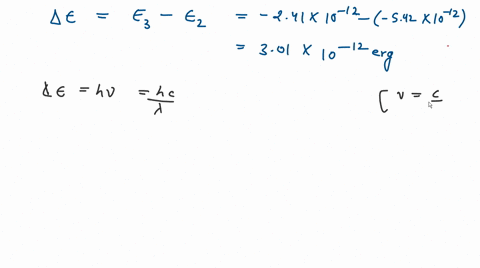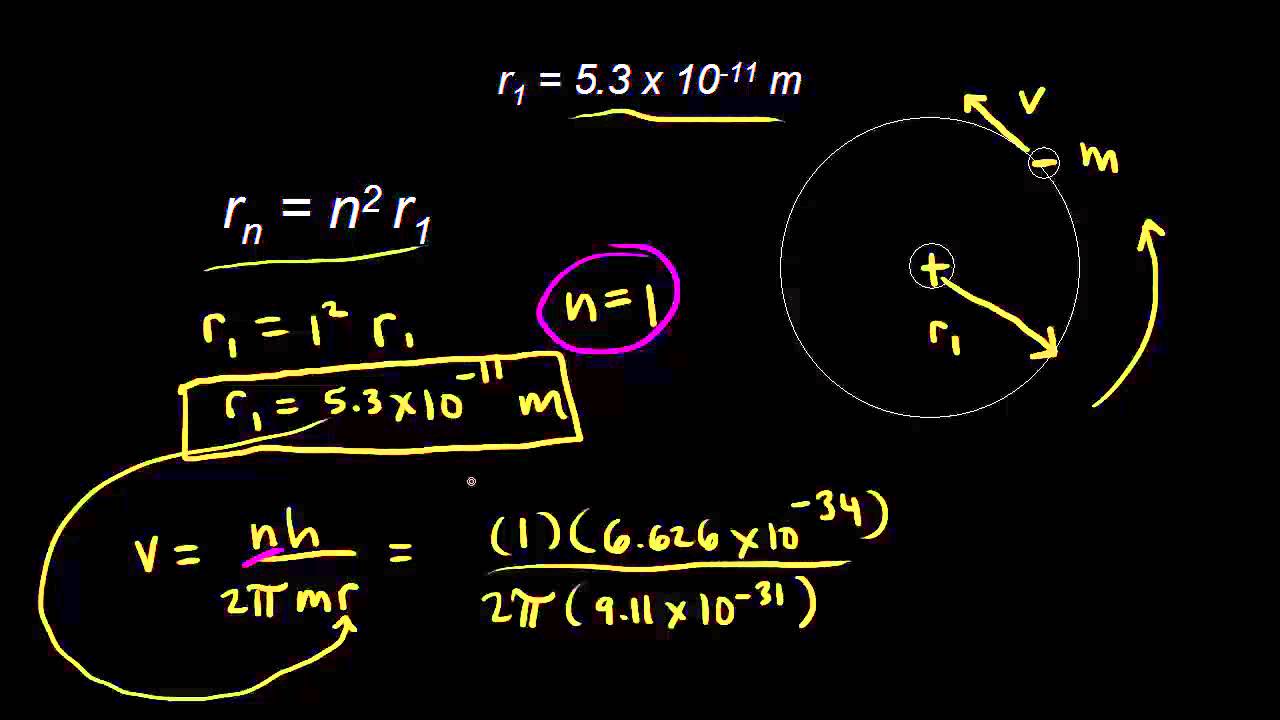![The kinetic energy of an electron in the second Bohr's orbit of a hydrogen atom is: [a0 is Bohr's radius] - Sahay Sir The kinetic energy of an electron in the second Bohr's orbit of a hydrogen atom is: [a0 is Bohr's radius] - Sahay Sir](https://sahay.guru/wp-content/uploads/2020/11/17ans-1024x515.jpg)
The kinetic energy of an electron in the second Bohr's orbit of a hydrogen atom is: [a0 is Bohr's radius] - Sahay Sir

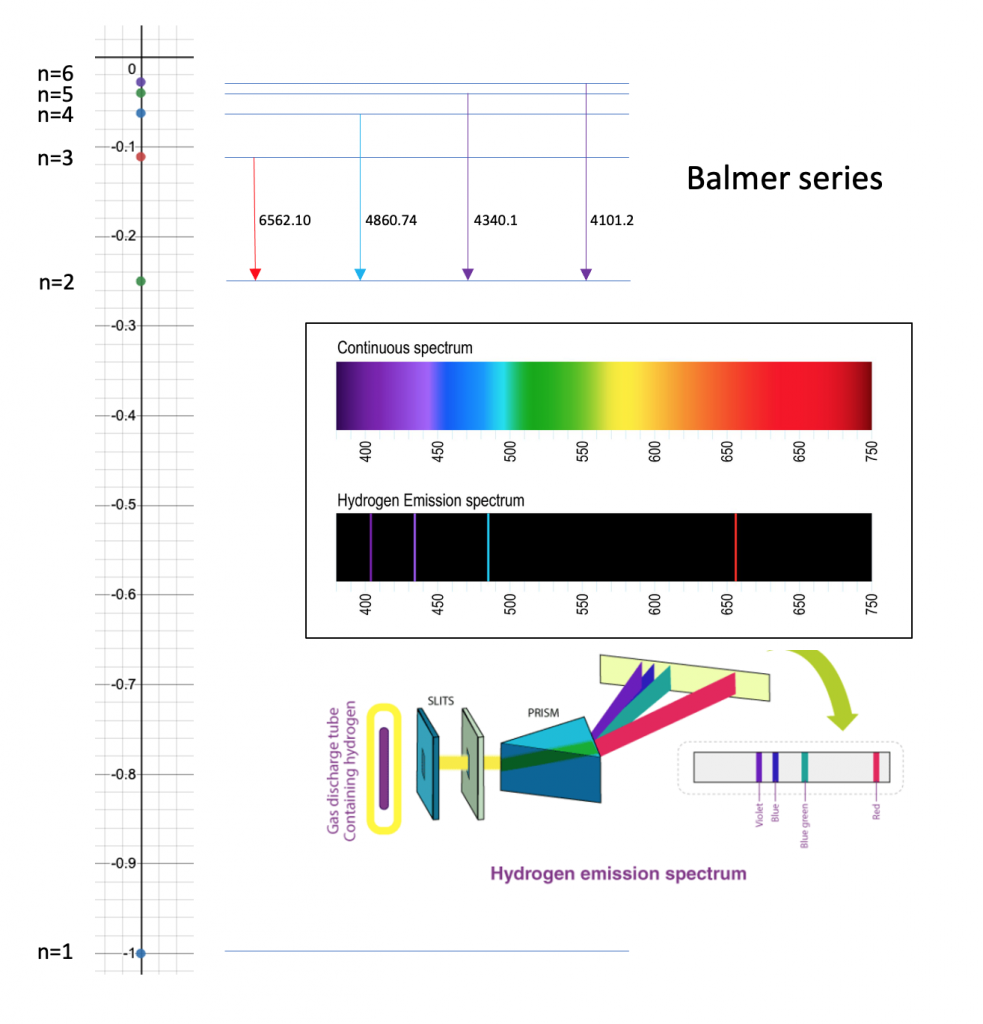
The energy of an electron in the first Bohr orbit for hydrogen is - 13.6 eV. Which one of the following is a possible excited state for electron in Bohr orbit of

Calculate the wavelength in Angstroms of the photon that is emitted when an electron is Bohr orb... - YouTube

3 Calculate the wavelength in angstrom of thephoton that is emitted when an electron inBohr orbit n=2 - Brainly.in

Bohr Model of the Atom. Experimental Observation of Hydrogen Line Emission In 1853, Anders Angstrom of Sweden first determined that a set of discrete. - ppt download